Clinical trials play an essential role in advancing the field of animal health. Through rigorous and methodical research, these trials contribute significantly to the development of effective treatments and preventive measures for a wide range of animal diseases. Given the intricate nature of veterinary medicine and the diverse species involved, conducting well-designed clinical trials is imperative to ensure safety and efficacy.
Clinical trials for animal health, much like those for human health, follow a series of well-defined stages, beginning from laboratory research to the application in real-world settings. This structured approach allows scientists to test the hypothesis of how a new drug, vaccine, or therapeutic procedure will perform in animals before its introduction to the market. Ensuring the robustness of these trials is critical for building trust among veterinarians, pet owners, and the broader community.
Key Insights
Key Insights
- Clinical trials are fundamental in establishing the efficacy and safety of new veterinary treatments.
- Randomized controlled trials (RCTs) in animals are a key technical consideration due to their ability to produce unbiased results.
- Implementing strict adherence to ethical guidelines and regulatory standards is crucial for actionable recommendations.
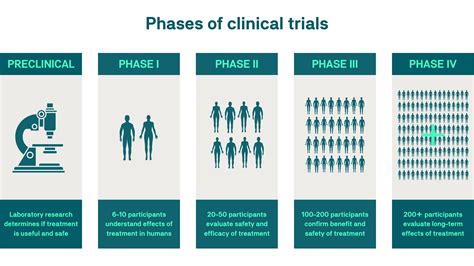
Stages of Clinical Trials in Animal Health
Clinical trials in animal health are segmented into several stages, paralleling the human clinical trial framework. Initially, preclinical studies involve laboratory experiments and initial testing in animal models to assess the safety and effectiveness of a treatment. These studies lay the groundwork for the subsequent phases. Once promising results are obtained, the trials advance to larger animal groups, where efficacy and side effects are evaluated more comprehensively.The phase I trials are akin to human clinical trials conducted on small groups to further test safety and optimal dosing. In veterinary medicine, this may involve several species since the treatment’s effects can vary significantly across animals. In phase II, the focus shifts to efficacy and optimal dosing schedules. This phase often uses larger groups and real-world settings to observe the treatment’s impact under varied conditions.
Finally, phase III trials involve extensive testing with a larger population to confirm the results observed in earlier phases. These trials provide critical data to regulatory bodies, which assess the balance between benefits and risks before granting approval for the treatment’s commercial use.
Ethical Considerations in Animal Clinical Trials
Conducting clinical trials on animals requires a meticulous approach to ethical considerations. Scientists must adhere to stringent guidelines to ensure humane treatment and minimize suffering. The ethical protocols involve ensuring that the benefits of the research justify any potential harm to the animals. This often includes the use of control groups, randomization, and blinding to reduce bias and ensure the validity of the results.Moreover, regulatory bodies like the FDA and EMA have set forth comprehensive guidelines to ensure the welfare of animals involved in these trials. Compliance with these guidelines is mandatory and includes the use of anesthesia where necessary, proper care, and humane endpoints. Implementing these practices ensures that the trials are both scientifically sound and ethically responsible, paving the way for the development of treatments that benefit both animals and, by extension, humans.
FAQ Section
What are the primary differences between clinical trials in animal health and human clinical trials?
While the overarching goals of clinical trials in both animal health and human medicine are to evaluate safety and efficacy, differences arise in the regulatory frameworks, ethical considerations, and species variability. Animal trials often involve more extensive ethical reviews and must follow strict welfare protocols, whereas human trials may encounter additional layers of regulatory scrutiny based on societal and legal standards.
How long do clinical trials in animal health typically take?
The duration of clinical trials in animal health varies based on the type and purpose of the trial. Preclinical studies can take months to years, while phases I, II, and III might collectively span several years. Regulatory approval and subsequent market introduction also add to the overall timeline.
In conclusion, clinical trials in animal health are a cornerstone of veterinary science, ensuring the development of safe and effective treatments. They adhere to stringent ethical and regulatory guidelines to safeguard animal welfare while providing invaluable data that enhances animal health outcomes.